The IATF and Its Purpose
The International Automotive Task Force (IATF) was formed to provide improved quality products to automotive customers worldwide. IATF members include 11 major vehicle manufacturers, such as BMW Group, Ford Motor Company, Stellantis and General Motors.[2]
Its primary purposes are to develop consensus on international fundamental quality system requirements, develop a common registration theme, provide training and support, and establish foreign liaisons with standards and regulatory bodies.[3]
IATF 16949:2016 is the international standard for automotive quality management systems. Its goal is the creation of a quality management system for continual improvement, greater defect prevention, and the elimination of supply chain waste.[4]
This principle, inherited from ISO 9001 and reinforced by IATF’s automotive additions, means the standard is focused on process effectiveness and efficiency.[5]
- Quality Improvement: Organizations report systematic defect prevention and reduction in variation after implementing IATF 16949.
- Cost Reduction: Reducing scrap, rework, warranty claims, and other wastes is a direct outcome of the standard’s focus on lean principles and variation reduction.
- Customer Satisfaction: Certification demonstrates to OEM customers that a supplier has robust quality systems.
- Global Market Access: Sites globally are certified, including a majority of the automotive supply base in regions like Europe, Asia, and the Americas.
- Supply Chain Trust: When all links in the chain (suppliers, subcontractors, etc.) are certified, OEMs have confidence that quality controls are in place throughout their supply network.
- Continuous Improvement Culture: Perhaps most importantly, IATF 16949 embeds a culture of ongoing optimization. Through its requirements, the standard pushes organizations to constantly identify weaknesses and drive improvements.
Top Nonconformities Under the New IATF 16949 Rules 6th Edition’s Rules
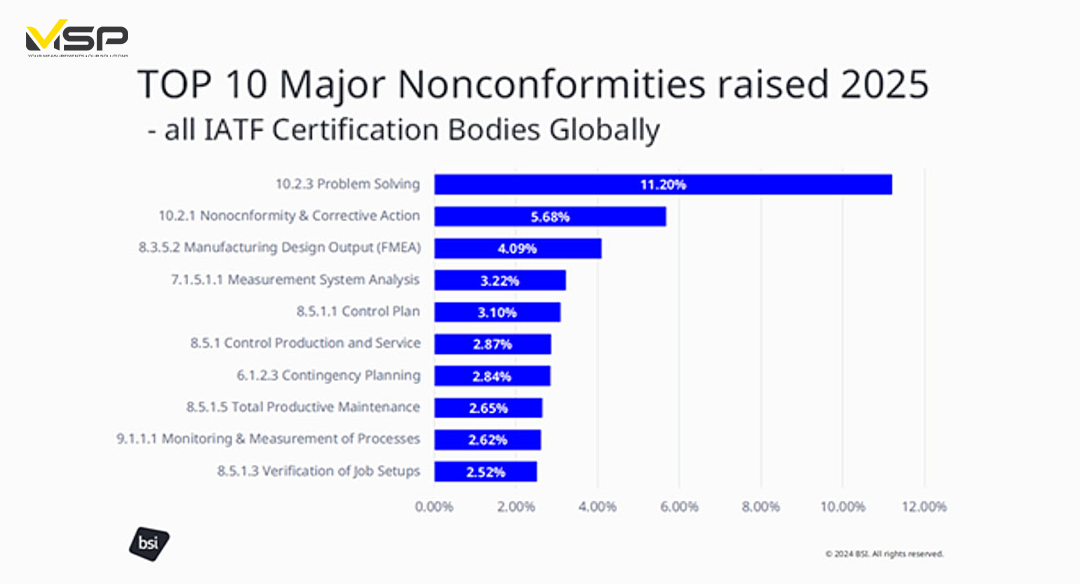
Certification bodies track and report the most common nonconformities (NCs) identified in IATF 16949 audits. Comparing recent data reveals some shifts in the “Top 10” issues, likely impacted by the new auditing focus, with the top major NCs cited across the globe in 2024 involving problem solving, nonconformity & corrective action, and manufacturing design output, respectively. Contingency planning remains a key concern (auditors are still finding gaps, though perhaps less severe ones). These results correlate with the Rule 6th focus on performance and effective corrective action. Data source: IATF www.iatfglobaloversight.org.
What is ISO 17025

ISO 17025 is an international standard that sets the guidelines for quality and competence in testing and calibration labs. It requires labs to follow high-quality processes and have a reliable Quality Management system to prove their expertise. ISO/IEC 17025 was developed through a collaboration between the International Organisation for Standardisation (ISO) and the International Electrotechnical Commission.
Ultimately, ISO 17025 Implementation is relevant to any organisation involved in testing, sampling, or calibration that aims to ensure the credibility of its results Simply put, if your work involves producing measurable outcomes, ISO 17025 likely applies to you. Even if your facility isn’t traditionally called a “lab,” under ISO 17020 Guide guidelines, it would be considered a laboratory if it produces quantifiable results.
Benefits of ISO 17025 Accreditation
Acquiring ISO 17025 accreditation provides numerous benefits when it comes to test and calibrate laboratories. Understanding the Benefits of ISO 17025 Accreditation is essential, as it enhances a laboratory’s credibility, improves operational efficiency, ensures accurate and reliable results, and boosts customer confidence. Here are some of the advantages:
1) Global Recognition
a) The ISO/IEC 17025 Certification ensures adherence to the highest quality standards in testing and calibration.
b) It guarantees reliable and accurate results for clients.
c) It solidifies the lab’s credibility in the global marketplace.
d) It significantly elevates the lab’s reputation as a trusted player internationally.
2) Consistent Data Standards
a) ISO 17025 makes sure that there’s uniformity in testing and calibration processes across all laboratories.
b) It guarantees reliable, repeatable results that meet global standards.
c) It facilitates easy comparison of data from different sources or labs.
d) It strengthens trust and credibility by maintaining consistent quality in data reporting.
3) Tender Eligibility
a) As ISO 17025 accreditation is recognised globally a lab can enjoy increased credibility when applying for tenders.
b) Once it oversees compliance with industry standards, it can your lab eligible for government and private sector contracts.
c) It strongly demonstrates commitment to quality and competence, which is often a requirement for tender eligibility.
4) Preferred Partner Status
a) ISO 17025 boosts reputation as a trustworthy lab, making you a preferred choice for clients.
b) It increases business opportunities by attracting clients who are keen on accredited and certified labs.
c) It provides confidence to stakeholders and makes sure they partner with a lab that meets international standards.
5) Competitive Advantage
a) ISO 17025 will help you differentiate your lab from competitors by showcasing better adherence to international standards.
b) It increases marketability and attracting more clients who seek certified, reliable results.
c) It improves efficiency and consistency, leading to higher quality outcomes compared to non-accredited labs.
Learn to implement ISO Methodologies with our ISO Certification Course today!
Key Requirements of ISO 17025
ISO 17025 sets forth several key requirements that laboratories must fulfil to achieve accreditation. These requirements ensure testing and calibration laboratories’ competence, impartiality, and consistent operation. The key requirements of 17025 are divided into the following:
a) General Requirements
It includes two factors – impartiality and confidentiality.
a) Impartiality implies that the laboratory must not prioritise commercial, financial or other benefits to compromise the quality of results.
b) Confidentiality makes sure the laboratory keeps the results and information private.
2) Structural Requirements
This involves employing personnel with appropriate qualifications, experience, and training. They must also ensure the use of validated test methods, proper calibration of equipment, and the availability of necessary resources.
3) Resource Requirements
This includes document control, personnel training, handling of non-conformities, and management review processes
4) Process Requirements
It involves activities such as quality assurance, sampling and reporting results. This ensures accuracy and comparability of test and calibration results.
5) Management System Requirements
These requirements are divided into two options, A and B. Option B will be applicable if a laboratory is part of a large organisation or has its own effective management. On the other hand, if the laboratory’s Quality Management system stays independent of any other management system, option A applies.
5) Management System Requirements
These requirements are divided into two options, A and B. Option B will be applicable if a laboratory is part of a large organisation or has its own effective management. On the other hand, if the laboratory’s Quality Management system stays independent of any other management system, option A applies.
What is the Certification Process for 17025 Accreditation?
The ISO 17025 Certification process involves several steps that laboratories must follow to obtain accreditation. Here’s an overview of the typical certification process:
Step 1: Familiarise yourself with the requirements of 17025. This includes:
a) Understanding the standard and its scope
b) Training all personnel about the management and technical activities
Step 2: Document laboratory activities. It involves documenting the following:
a) Policies
b) Procedures
c) Work instructions
Step 3:Identify areas for improvement and make sure that the laboratory’s processes are functioning effectively.
Step 4: Review the preparedness of your organisation. Before applying for accreditation, your laboratory must meet the accreditation body’s requirements. It means that you need to possess the following:
a) All necessary documents
b) Internal audit programme
c) Records of every management review and method validation.

Relevance for MSP Metrology & Roughness Testing
Given what IATF 16949 and ISO/IEC 17025 require:
-
MSP Metrology’s roughness-tester services — if performed under a lab accredited (or compliant) to ISO/IEC 17025 — can serve as a valid calibration/inspection service in the supply chain.
-
This alignment means MSP Metrology can be positioned as a trusted partner for automotive-industry clients who need to meet IATF 16949 requirements — offering measurement traceability, reliable surface-finish inspection, and documentation.
-
In marketing or proposals, emphasising that MSP offers ISO/IEC 17025-based testing/calibration (especially roughness testing) becomes a strong selling point to automotive suppliers concerned with both product quality and compliance.
For More Info Us, Visit Our Socials
MSP Metrology
Your Measurements, Our Solutions.
See also our services: https://mspmetrology.com/msp-services/
Above sharing are for our latest announcement and news. If you have any enquiry with any other metrology products, welcome to contact us by email: enquiry@mspmetrology.com



